
Medical device consulting helps companies fill critical expertise gaps across product development, design controls, risk management, regulatory readiness, and quality systems. For teams facing tight timelines, remediation work, or complex programs such as combination products, the right consulting partner can reduce delays, strengthen documentation, and provide access to specialized technical talent when it matters most.
Medical device companies rarely need support in just one area. A growing team may need help defining user needs, planning verification and validation, updating a risk management file, remediating a quality system, or adding temporary project resources at the same time. Ventura Solutions’ service model reflects that reality by combining consulting, integrated solutions, staffing, and training for medical device and pharmaceutical organizations.
What medical device consulting includes
Medical device consulting is broader than regulatory advice alone. In practice, it can include product development guidance, design control deliverables, risk management activities, quality system support, project management, regulatory support, and technical problem-solving across different stages of the product lifecycle. Ventura Solutions’ current consulting and integrated solution pages highlight services in design controls, risk management, design and development, quality assurance, regulatory affairs, DHF remediation, and end-to-end product development.
For example, design controls remain a core part of medical device development. FDA guidance explains that design controls are intended to build quality, safety, effectiveness, and savings into medical devices, while Ventura Solutions’ design controls page lists support for development plans, user needs, design inputs, design outputs, verification, validation, design reviews, trace matrices, and design transfer.
Risk management is another major consulting area. FDA materials describe risk management as a structured, risk-based activity within a quality system, and ISO 14971 defines a process for medical device risk management. Ventura Solutions specifically states that it supports ISO 14971 deliverables such as risk management plans and reports, DFMEA, PFMEA, hazard analysis, use-related risk analysis, clinical risk-benefit analysis, and risk management files.
That is why medical device consulting is often most valuable when a company does not just need advice, but needs expert help producing the actual work: documents, analyses, reviews, remediation plans, and cross-functional execution.
When companies should hire a medical device consulting partner
The best time to bring in a consulting partner is usually before a bottleneck becomes a major delay. Many companies seek outside support when internal teams are overloaded, when a new product program requires specialized expertise, or when quality and regulatory documentation needs to be corrected quickly. Ventura Solutions’ About page says its primary focus areas include design history file and quality system remediation, quality and regulatory support, and product development services for the medical device and pharmaceutical industry.
You may need medical device consulting when:
- Your team lacks specialized expertise in design controls, risk management, or regulatory support
- A product program is moving faster than internal bandwidth allows
- Documentation gaps are slowing development or audit readiness
- You need support for remediation, traceability, or quality system implementation
- Your program includes drug-device complexity and requires combination products consulting
- You need expert support now while building a longer-term hiring plan
For startups and scaling manufacturers, the benefit is not only technical support. It is also flexibility. Ventura Solutions notes that it can manage product development from concept to launch or be “pieced in as needed,” which is often exactly what growing teams need when budgets, timelines, and internal capabilities are still evolving.
Medical device consulting vs medical device staffing
Medical device consulting and medical device staffing solve related but different problems. Consulting typically delivers specialized knowledge, technical leadership, documentation support, and project-based guidance. Staffing, by contrast, helps companies add people to fill capability gaps or increase execution capacity. Ventura Solutions separates these offers on its site, with consulting and integrated solutions on one side and niche staffing and recruiting for medical device and pharmaceutical companies on the other.
In many cases, companies need both. A consultant may define the strategy, structure the work, or lead a remediation effort, while contract or permanent hires provide the bandwidth to execute the workload. Ventura Solutions’ staffing pages say the company specialises in solving challenging resource needs and uses scientific and engineering managers in the recruitment process, with optional training support as well.
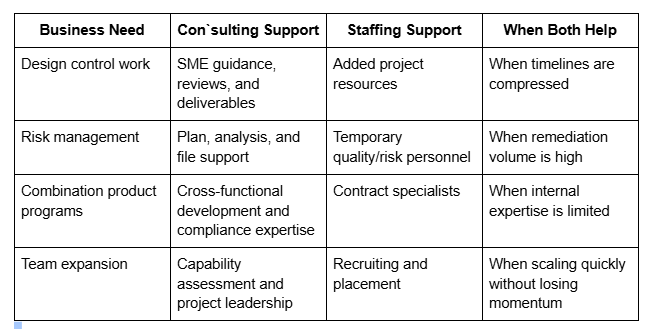
This distinction also helps buyers choose the right model. If the main issue is lack of direction or technical depth, consulting is often the better first step. If the issue is lack of manpower, staffing may be the immediate need. If the issue is both, a partner that can support consulting and staffing is often more efficient.
Why combination products consulting deserves special attention
Combination products add complexity because they combine two or more regulated components, such as drug-device, biologic-device, or drug-device-biologic combinations. FDA explains that combination products may be physically combined into a single entity or packaged together as combined regulated products, and 21 CFR Part 4 establishes how current good manufacturing practice requirements apply to them.
That complexity is one reason combination products consulting deserves its own discussion. These programs often require closer alignment across design controls, risk management, traceability, quality systems, and regulatory strategy. Ventura Solutions positions combination products as a dedicated consulting service and also offers an end-to-end integrated device solution for clients managing the device side of combination product development.
Ventura Solutions own recent content on traceability notes that traceability connects requirements, design, verification, validation, and post-market data into a single auditable system of evidence in combination product development. That makes outside support especially valuable when teams are coordinating across multiple disciplines and document sets.
In practical terms, combination products consulting can help teams:
- Align technical development with compliance expectations
- Improve traceability across risks, requirements, and testing
- Reduce confusion between device and drug-related responsibilities
- Strengthen project coordination across engineering, quality, and regulatory functions
- Keep documentation moving as complexity increases
What to look for in a medical device consulting company
Not all consulting firms bring the same type of value. In a regulated industry, the best partner is not just a general adviser but a team that understands development, documentation, risk, quality, and execution. Ventura Solutions’ site emphasises technical depth in design controls, risk management, product development, quality systems, and combination products, while client testimonials highlight both technical expertise and communication skills.
When evaluating a consulting partner, look for:
- Direct experience in medical device or combination product development
- Fuency in design controls, risk management, and quality systems
- Ability to contribute to actual deliverables, not only high-level advice
- Strong cross-functional communication skills
- Flexible engagement models, including project support, integrated teams, or staffing
- Training capability to strengthen internal teams over time
A strong partner should also help your team become more effective, not more dependent. Ventura Solutions’ training offerings in design controls, risk management, and regulatory compliance suggest a model that supports both immediate project needs and longer-term capability building.
How expert consulting can reduce delays and strengthen execution
Medical device development slows down when teams lack clarity, capacity, or specialized expertise. Documentation gets delayed. Reviews take longer. Risks are found late. Development decisions become harder to defend. This is where expert consulting can create measurable value: better structure, faster progress, stronger documentation, and fewer avoidable rework cycles. FDA’s design control and risk management guidance also reinforces the importance of building quality and risk-based decision-making into the development process rather than trying to fix gaps later.
For many companies, the goal is not simply to “outsource” work. It is to gain access to the right expertise at the right time. Ventura Solutions frames its business around helping clients accelerate product development and ensure market compliance, which is exactly the outcome many medical device teams are trying to achieve when they seek consulting support.
FAQs About Medical Device ConsultingWhat does a medical device consulting company do?
A medical device consulting company helps with specialized areas such as product development, design controls, risk management, quality systems, regulatory support, remediation, and technical project execution. Ventura Solutions also extends this support through integrated solutions, staffing, recruiting, and training.
When should a company hire a medical device consultant?
Companies typically hire consultants when they face expertise gaps, aggressive timelines, remediation work, audit or documentation pressure, or complex programs that require outside support. Ventura Solutions’ service pages strongly align with these use cases.
What is the difference between medical device consulting and medical device staffing?
Consulting provides technical expertise, strategic guidance, and deliverable support. Staffing provides people to fill resource gaps. Some companies need one, while others benefit from both at the same time.
What is combination products consulting?
Combination products consulting focuses on programs that combine regulated components such as drugs, devices, and biologics. FDA regulates these products under a distinct framework, and they often require stronger coordination across design, risk, quality, and compliance functions.
Can consulting help with risk management and design controls?
Yes. FDA guidance and ISO 14971 both support a structured approach to risk management and development controls, and Ventura Solutions specifically lists deliverables in both areas, including design plans, trace matrices, DFMEA, PFMEA, hazard analysis, and risk management files.
Can a consulting company also help with staffing for medical device companies?
Some can. Ventura Solutions is a clear example because it offers both consulting services and niche staffing and recruiting for medical device and pharmaceutical companies.
What should companies look for in a consulting partner?
Look for technical depth, relevant industry experience, strong communication, flexible engagement models, and the ability to contribute to actual project deliverables. Testimonials and service-page specificity can help validate that expertise.
The Value of Medical Device Consulting Across Development, Compliance, and Staffing
Medical device consulting is most valuable when it helps companies move from uncertainty to execution. Whether the need is design controls, risk management, quality system support, combination products consulting, or medical device staffing, the right partner should bring both technical credibility and practical momentum. Ventura Solutions’ current positioning as a consulting, staffing, recruiting, and training partner for medical device and pharmaceutical companies makes it well aligned to support organizations that need flexible, specialized help across the product lifecycle.
